 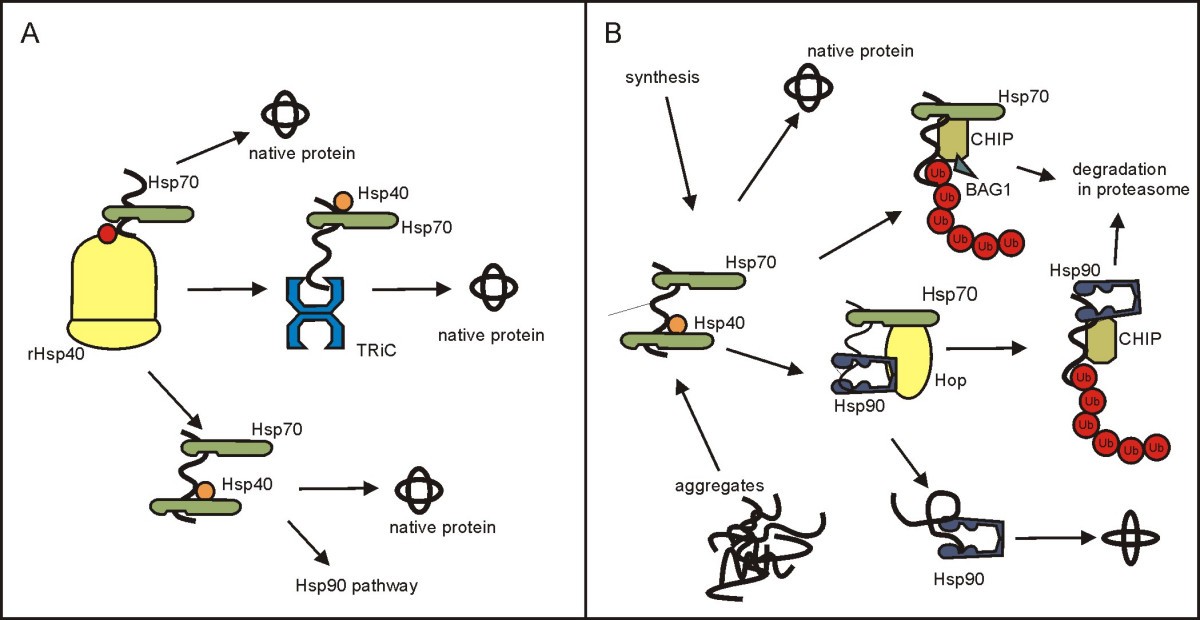
Targeting to specific locations within a cellular compartment and binding of specific substrates for delivery to Hsp70 have been identified as modes of J-protein specialization. In addition, J-proteins drive the functional diversity of Hsp70 chaperone systems through action of regions outside their J-domains. All J-protein co-chaperones play the essential role, via action of their J-domains, of stimulating the ATPase activity of Hsp70, thereby stabilizing its interaction with substrate. 
Īli MM, Roe SM, Vaughan CK, Meyer P, Panaretou B, Piper PW, Prodromou C, Pearl LH (2006) Crystal structure of an Hsp90-nucleotide-p23/Sba1 closed chaperone complex.By binding to a multitude of polypeptide substrates, Hsp70-based molecular chaperone systems perform a range of cellular functions. Īlderson TR, Roche J, Gastall HY, Dias DM, Pritišanac I, Ying J, Bax A, Benesch JLP, Baldwin AJ (2019) Local unfolding of the HSP27 monomer regulates chaperone activity. Īlbakova Z, Armeev GA, Kanevskiy LM, Kovalenko EI, Sapozhnikov AM (2020) HSP70 multi-functionality in cancer. Īkerfelt M, Morimoto RI, Sistonen L (2010) Heat shock factors: integrators of cell stress, development and lifespan. The Author(s), under exclusive license to Springer Nature Switzerland AG.Īcharya P, Kumar R, Tatu U (2007) Chaperoning a cellular upheaval in malaria: heat shock proteins in Plasmodium falciparum. Hsp110 Hsp60 Hsp70 Hsp90 J domain protein Molecular chaperone. Members of the main classes of heat shock proteins are all represented in Plasmodium falciparum, the causative agent of cerebral malaria, and they play specific functions in differentiation, cytoprotection, signal transduction, and virulence. Vector-borne parasites affecting human health encounter stress during transmission between invertebrate vectors and mammalian hosts. Their function is particularly important in disease due to increased stress in the cell. The general structural and functional features of the major heat shock protein families are discussed, including their roles in human disease. Heat shock proteins do not function in isolation but are rather part of the chaperone network in the cell. Most heat shock proteins form large oligomeric structures, and their functions are usually regulated by a variety of cochaperones and cofactors. Heat shock proteins are localised to different compartments in the cell to carry out tasks specific to their environment. The large and varied heat shock protein class is categorised into several subfamilies based on their sizes in kDa namely, small Hsps (HSPB), J domain proteins (Hsp40/DNAJ), Hsp60 (HSPD/E Chaperonins), Hsp70 (HSPA), Hsp90 (HSPC), and Hsp100. Heat shock proteins are induced by a variety of stresses, besides heat shock. This is achieved by ATP-dependent (folding machines) or ATP-independent mechanisms (holders). 
Heat shock proteins form the major class of molecular chaperones that are responsible for protein folding events in the cell. They play crucial roles in facilitating the correct folding of proteins in vivo by preventing protein aggregation or facilitating the appropriate folding and assembly of proteins. Molecular chaperones are a group of structurally diverse and highly conserved ubiquitous proteins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
